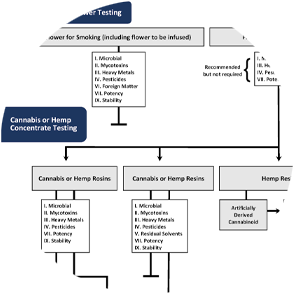
Required Testing Flow chart
Download this flow chart which shows something dolor ipsum unum set eplurubus unum Download this flow chart which shows something dolor ipsum unum set eplurubus unum
Sample Size Requirements
Minnesota regulations require that samples represent 0.5% of the total batch mass. The minimum sample size varies based on your batch size and must include both a compliance sample (for laboratory testing) and a retention sample (to be held by the licensee).

Important: Retention Samples
Licensees must retain a 23 g portion of each representative sample for six months after the product expiration date. This retention sample must be stored appropriately for the product matrix (refrigerated for perishables, room temperature for shelf-stable items).
Chain of Custody
Maintaining proper chain of custody documentation is critical for regulatory compliance and sample integrity. When submitting samples to Analytica Inc., you must:
- Schedule testing through the statewide monitoring system within two business days of sampling
- Complete our laboratory chain of custody form with all required information
- Document sample transfer from your facility to our laboratory
- Ensure samples are transported securely with appropriate temperature control
- Verify sample receipt with our laboratory staff
All chain of custody documentation is maintained by Analytica Inc. and recorded in the statewide monitoring system for regulatory traceability.
Special Considerations
Flower for Concentrate Production
If flower is intended for further processing into concentrates rather than direct retail sale, batch testing is not required under Minnesota’s technical standards. However, both Analytica and the OCM strongly recommend testing for microbial contaminants, heavy metals, pesticides, and potency before extraction, as these contaminants can be concentrated during processing.
Manufactured Final Products
Homogeneity and stability testing must be conducted on products in their final packaged form. For homogeneity testing, provide 10 individual units randomly selected from the batch, including units from multiple packages when applicable.
Stability Testing Samples
Stability testing requires samples at multiple time points. Provide sufficient sample volume in final packaging to allow testing at each designated time point throughout the product’s intended shelf life.
Pre-Submission Checklist
Before submitting samples to Analytica Inc., verify that you have:
- Collected the required sample size (See table)
- Followed proper sampling procedures with sterilized equipment
- Properly labeled all sample containers with batch information
- Set aside the required retention sample for storage at your facility
- Scheduled testing in the statewide monitoring system
- Completed the laboratory chain of custody form
- Arranged appropriate transport with temperature control if needed
- Provided complete pesticide/solvent disclosure information when applicable
